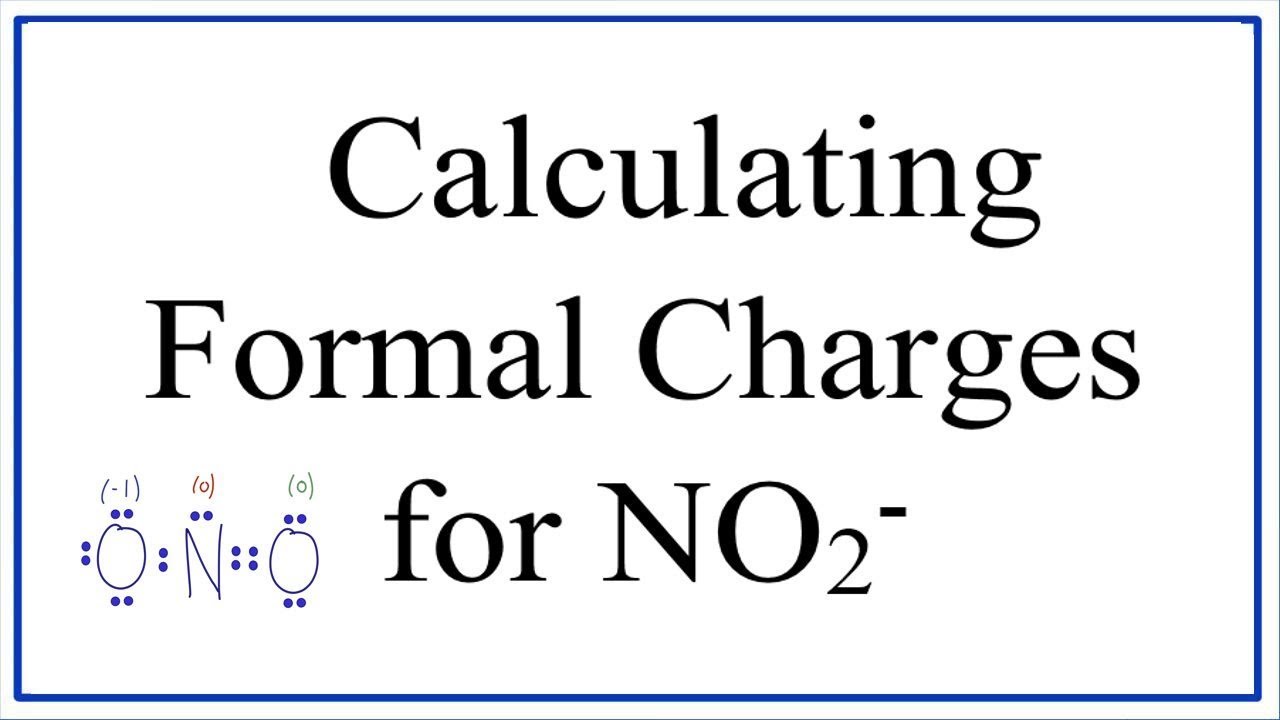
The geometry of Molecules is a platform where we try to educate you about various chemistry concepts and find the answers you have been looking for. And it might seem challenging, but it becomes the most fun and easy subject to learn if learned the correct way. Determine the formal charge on each atom in the following molecules or ions: (a) N2H4 (b) PO43 (c) BH4 (d) NH2OH arrowforward Compute the formal charges on the atoms in the followingLewis diagram, which represents the azide ion (N3 ). Sounds easy, right? Go ahead and try our formal charge calculator.ĭo you know that a perfect diamond is made up of a single Carbon molecule? Chemistry is one of the most exciting subjects. If there is more than one possible Lewis structure, choose the one most likely preferred. Draw the Lewis Structure for the following molecules and ions and calculate their formal charge. And to make it easy for you to do the calculations we have developed a Formal charge calculator for you, where you have to just enter the number of valence electrons, non-bonding electrons, and bonding electrons to find out the formal charge. Draw a Lewis structure (including all lone pair electrons) and calculate the formal charge (FC) of each atom of nitrosyl chloride (ClNO) FC on Cl FC on N FC on O. Similarly, you can use this formula for other molecules and find out formal charges for individual atoms in the molecule. And using the formula stated above, we found out that the total charges on both Sulphur and Fluorine atoms are zero. Oxide Nitrous oxide is a nitrogen oxide with an N2O chemical formula. 
Here in SF4 as all the Fluorine atoms are arranged symmetrically, they will have the same charges. Show 1 day ago The Lewis structure of hydrogen and 2 oxygen atoms shows a total. A: Click to see the answer Q: SOCl2, thionyl chloride: Calculate formal charges for each atom. of valence electrons Non-bonding electrons Bonding electrons Total charge Sulphur (S) Now that we know the formula let us look at the example of how to find out formal charges for individual atoms in a polyatomic molecule Let us calculate the formal charges of SF4 Atom Total no. One can calculate the formal charges for any given atom with the help of the following formula:į.C = Valence electrons – Nonbonding electrons- Bonding electrons/2 These charges help in knowing if the given structure of the molecule is stable or not. So, the final answer is $\textbf$.Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. With formal charges of +1 on N1, -1 on N2, and -1 on O. Now the formal charges are minimized, and the most likely Lewis structure for nitrous oxide is: Recalculate the formal charges for the adjusted structure.įormal charge = 5 - 1 - 0.5(6) = 5 - 1 - 3 = 1įormal charge = 5 - 3 - 0.5(6) = 5 - 3 - 3 = -1 
The formal charges are not minimized, so we can adjust the structure by moving one lone pair from N1 to form a double bond with N2.Ħ. N2O calculate the formal charges on each atom below. Two Lewis structures are given below for dinitrogen monoxide. FC(H) (1 valence electrons) (0 lone pair electrons) 1 2(2 bonding electrons) 0. Using Equation 2.3.1 to calculate the formal charge on hydrogen, we obtain. Each hydrogen atom in the molecule has no non-bonding electrons and one bond. Check if the formal charges are minimized and adjust the structure if necessary. Answer We are given the two Lewis dot structure of N2O and the formal charge of each element in each structure as follow I)N2O We know the formula for the formal charge Formal charge. A neutral hydrogen atom has one valence electron. Calculate the formal charges for each atom.įormal charge = (valence electrons in free atom) - (nonbonding electrons) - 0.5(bonding electrons)įormal charge = 5 - 3 - 0.5(4) = 5 - 3 - 2 = 0įormal charge = 6 - 6 - 0.5(2) = 6 - 6 - 1 = -1ĥ. Test yourself Determine the formal charge for each atom in NCl 3. This gives the formal charge:Br: 7 7 0Cl: 7 7 0All atoms in BrCl 3 have a formal charge of zero, and the sum of the formal charges totals zero, as it must in a neutral molecule. Calculate the formal charges for each of the oxygen atoms within the nitrite ion, NO2. Subtract this number from the number of valence electrons for the neutral atom. We will add 6 electrons to O (3 lone pairs) and 3 electrons to each N atom (1.5 lone pairs).Ĥ. Be the first to ask a question about this topic. We have used 4 electrons for the N-N and N-O bonds, so we have 12 electrons left to distribute. 
Distribute the remaining valence electrons as lone pairs. Since there are 2 N atoms and 1 O atom, the total number of valence electrons is 2(5) + 6 = 16.ģ. N has 5 valence electrons and O has 6 valence electrons. Determine the total number of valence electrons in the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
